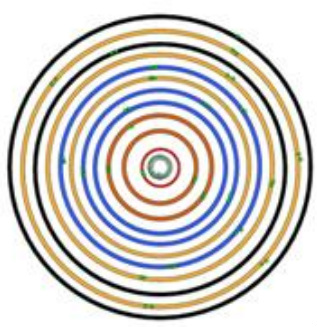
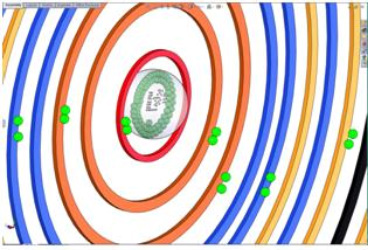
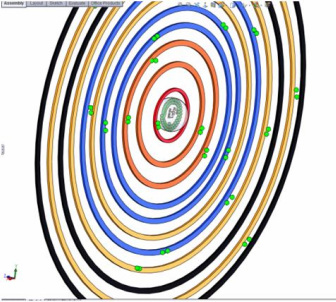
SolidWorks Model: An Indium Atom
After, I have completed the research on the Indium atom I begin to build the model. The model must have the number of neutrons, protons, and electrons required for that atom. Each energy level must be a different color so the overlapping sublevels can be seen. Each sublevel needs the correct number of paired or unpaired electrons.
Indium's atomic number is 49, so the atom has 49 protons, 66 neutrons, and 49 electrons. The 49 electrons are distributed between 5 energy levels.
Indium's atomic number is 49, so the atom has 49 protons, 66 neutrons, and 49 electrons. The 49 electrons are distributed between 5 energy levels.
Skills
For this worksheet I used a mixture of Chemistry skills and SolidWorks skills
Chemistry Skills
· Distinguish among principal energy level, energy sublevel, and atomic orbital
· Apply the Aufbau principle, the Pauli exclusion principle, and Hund's rule to write the electron configurations of the elements
· Distinguish between metals, nonmetals, metalloids
· Distinguish between a group and a period.
Design processes
· Adding parts to an assembly
Chemistry Skills
· Distinguish among principal energy level, energy sublevel, and atomic orbital
· Apply the Aufbau principle, the Pauli exclusion principle, and Hund's rule to write the electron configurations of the elements
· Distinguish between metals, nonmetals, metalloids
· Distinguish between a group and a period.
Design processes
· Adding parts to an assembly
Justification
I chose this project because I had to use all of the skills listed to research and create this atom model. Also, I chose this project to show my knowledge on electron orbital filling diagrams.
Skills such as adding parts to an assembly can help me later, not only to help me further my Chemistry knowldge, but help me become more familiar with SolidWorks.
Skills such as adding parts to an assembly can help me later, not only to help me further my Chemistry knowldge, but help me become more familiar with SolidWorks.